Department of Physical Chemistry
Soft Matter Group
Short URL for media go.spbu.ru/rgvictorov
Group Members
 |
Group Leader Alexey I. VictorovDoctor of Chemical Sciences, Professor, Head of Department of Physical Chemistry https://orcid.org/0000-0001-9994-1297 This email address is being protected from spambots. You need JavaScript enabled to view it. room 2122 |
 |
Alexandr A. Vaninassociate professor, PhD This email address is being protected from spambots. You need JavaScript enabled to view it. room 2168 |
 |
Andrey Yu. Vlasovassociate professor, PhD This email address is being protected from spambots. You need JavaScript enabled to view it. room 2166 |
 |
Igor Yu. Gotlibassociate professor, PhD This email address is being protected from spambots. You need JavaScript enabled to view it. room 2152 |
 |
Evgenia A. Safonovaassociate professor, PhD https://orcid.org/0000-0002-8660-0909 This email address is being protected from spambots. You need JavaScript enabled to view it. rooms 2162, 2145 |
 |
Yuri G. Dobrjakovengineer, PhD This email address is being protected from spambots. You need JavaScript enabled to view it. rooms 2164, 2161 |
 |
Alina S. Konevaengineer This email address is being protected from spambots. You need JavaScript enabled to view it. room 2162 |
 |
Petr A. KorchakPhD student, engineer https://orcid.org/0000-0001-8818-5748 This email address is being protected from spambots. You need JavaScript enabled to view it. rooms 2153 |
 |
Polina Sorinamaster student, research assistant This email address is being protected from spambots. You need JavaScript enabled to view it. room 2152 |
 |
Kristina NikiforovaPhD student This email address is being protected from spambots. You need JavaScript enabled to view it. |
Alumni
 |
Ksenia A. EmelyanovaPhD |
 |
Ekaterina Yakovlevamaster degree, 2021 |
 |
Veronika Fedotovamaster degree, 2021 |
 |
Pavel Ivanovmaster degree, 2021 |
 |
Mikhail Karnaukhbachelor degree, 2020 |
 |
Alexandra NikolaevaPhD, 2017 |
 |
Anastasia VenediktovaPhD, 2013 |
Research Area
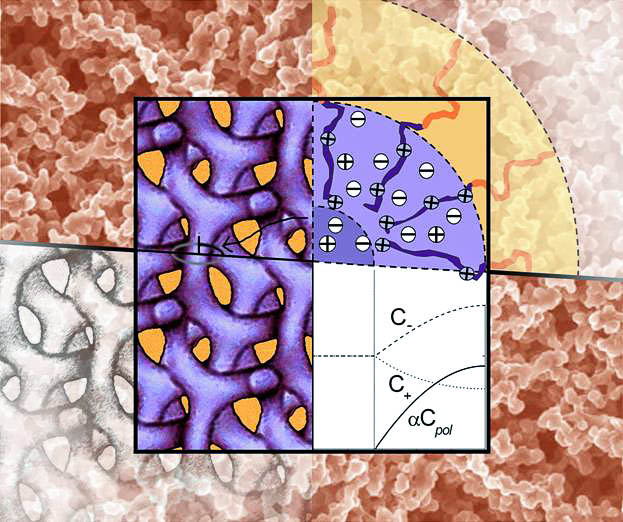
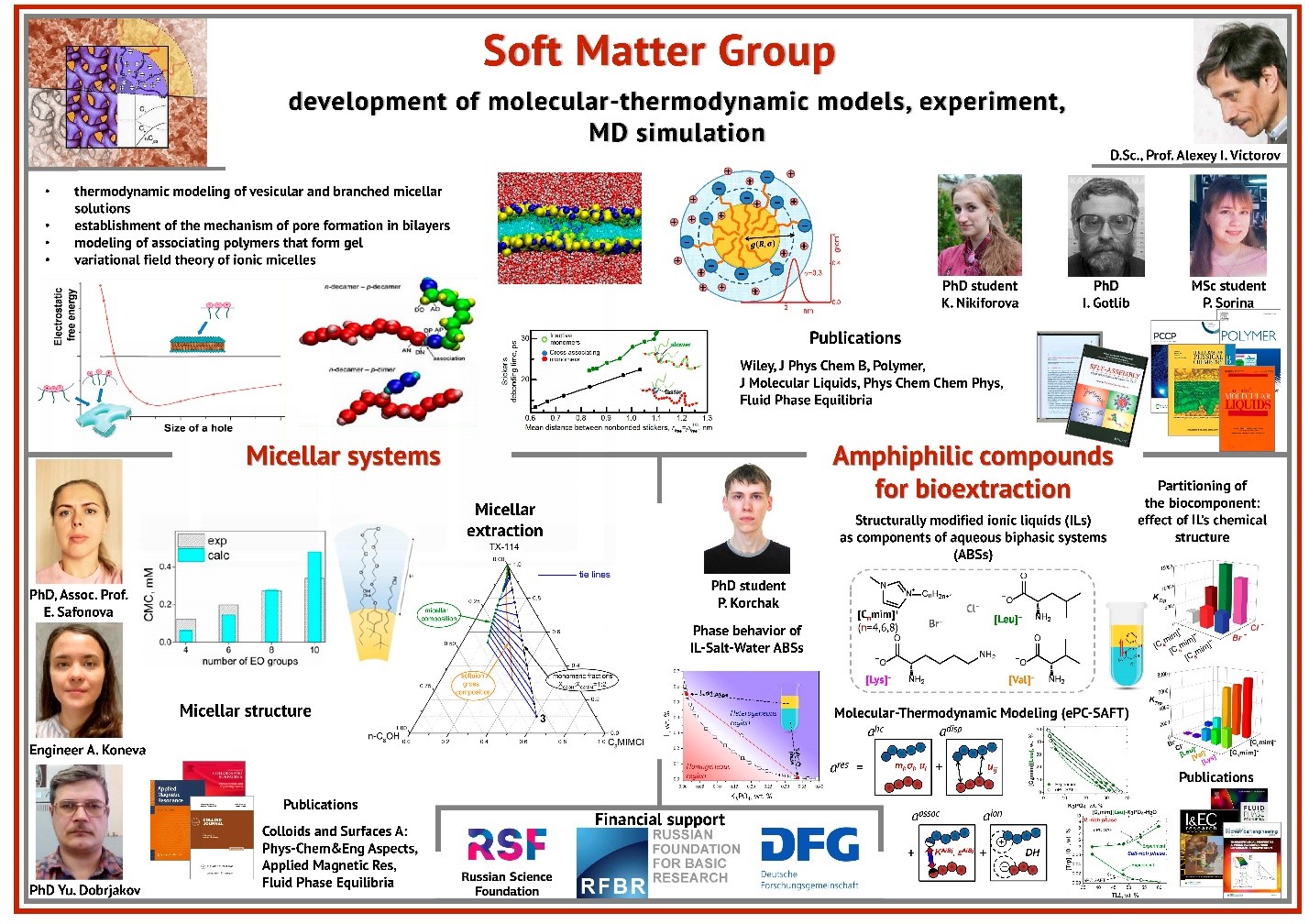
Amphiphilic molecules may self-assemble, forming rich variety of structures. For fluid systems, a minor change of the environment may cause radical structural changes. Such soft mesoscale structures are found in many systems, including solutions of classical surfactants, block copolymer solutions and melts, biologic membranes, etc. Structural rearrangements often result in a crucial change of macroscopic properties (volume and elasticity of the sample, its viscosity, selectivity with respect to different solvents and salts, electric conductivity and so on). This can be used in the design of environmentally responsive smart materials whose behavior may be manipulated by external stimuli. The role of such materials is rapidly increasing in various applications. The major problem is to relate the macroscopic behavior of a complex fluid system with its molecular structure and to develop predictive models for engineering applications. We perform experimental studies and develop molecular-thermodynamic models for different types of complex fluids.
Funded projects
- RSF “Distribution of biocomponents in mesoscopic fluids that contain amphiphilic molecules of differing structure”, head: Prof. A.I. Victorov, 2020-2023.
- RFBR “Effect of nanoscale organization on the shift of chemical equilibria in mesostructural fluid systems: the development of molecular-thermodynamic theory and computer simulation”, head: Prof. A.I. Victorov, 2018-2020.
- RFBR “ХХII International Conference on Chemical Thermodynamics in Russia RCCT-2019”, head: Prof. A.I. Victorov, 2019.
- RFBR-DFG “Mixed micellar systems in continuous reactive couter-current extraction columns for biocatalytic reactive separation”, head: Ass. Prof. E.A. Safonova, Apr. 2016 – Apr.2019.
Selected Publications
Book chapter
Victorov A., Self-Assembly into Branches and Networks, Chapter 2, pp 41-76. In: Self -Assembly: From Surfactants to Nanoparticles, Ed. R.Nagarajan, Wiley Series on Surface and Interfacial Chemistry (NY),1st Edition, January 2019, 400 Pages, Hardcover, Wiley & Sons Ltd , ISBN: 978-1-119-00136-2
Papers
- Iakovleva, Ekaterina A.; Sorina, Polina O.; Safonova, Evgenia A.; Victorov, Alexey I. Specific Interactions in the Model of Mixed Multicomponent Micelles: Predicting Aggregation Behavior and Details of Structure. Fluid Phase Equilibria (2022) 556, 113376 https://doi.org/10.1016/j.fluid.2022.113376
- Iakovleva, Ekaterina A.; Safonova, Evgenia A.; Victorov, Alexey I. Aggregates of Ethoxylated Surfactant with Added n-Octanol: Details of Corona Structure from a Molecular-Thermodynamic Model. Fluid Phase Equilibria (2021) 546, 113134 https://doi.org/10.1016/j.fluid.2021.113134
- Korchak, Petr A.; Safonova, Evgenia A.; Victorov, Alexey I. Partitioning of L-Tryptophan in Aqueous Biphasic Systems Containing an Alkylimidazolium Ionic Liquid and a Phosphate Salt. Industrial and Engineering Chemistry research (2021) 60, № 44, 16078–16088 https://doi.org/10.1021/acs.iecr.1c02178
- Budkov, Y.A., Victorov, A.I. Simple analytical theory for micelles with widespread radial distribution of charged heads Journal of Molecular Liquids (2021) 341, 117438 https://doi.org/10.1016/j.molliq.2021.117438
- Emelyanova, K.A., Sorina, P.O., Victorov, A.I. Transmembrane potential in vesicles formed by catanionic surfactant mixtures in an aqueous salt solution. Physical Chemistry Chemical Physics (2020) 22(45), 26438–26451 https://doi.org/10.1039/D0CP05248E
- Mohammad Izadyar, Mojtaba Rezaeian and Alexey Victorov. Theoretical Study on the Absorption of Carbon dioxide by DBU-based Ionic Liquids. Phys. Chem. Chem. Phys. (2020), 22, 20050-20060 https://doi.org/10.1039/D0CP03612A
- Sofia V. Koroleva, Petr Korchak, Alexey I. Victorov. Molecular Thermodynamic Modeling of the Specific Effect of Salt on the Aggregation of Nonionic Surfactants. Journal of Chemical & Engineering Data (2020) 65, 987−992 https://doi.org/10.1021/acs.jced.9b00303
- Igor Yu. Gotlib, Alexey I. Victorov. Association kinetics and equilibrium in solutions of cross-associating chains that contain inactive spacers. Polymer (2020) 187, 122085 https://doi.org/10.1016/j.polymer.2019.122085
- Korchak, P. A., E. V. Alopina, I. B. Pukinsky, and E. A. Safonova. Liquid-Liquid Equilibria of Aqueous Biphasic Systems Containing 1-Alkyl-3-Methylimidazolium Amino Acid Ionic Liquids with Different Anions (L-Leucine, L-Valine, L-Lysine) and Inorganic Salt (Tripotassium Phosphate, Potassium Carbonate). Fluid Phase Equilibria (2020) 525, 112789 https://doi.org/10.1016/j.fluid.2020.112789
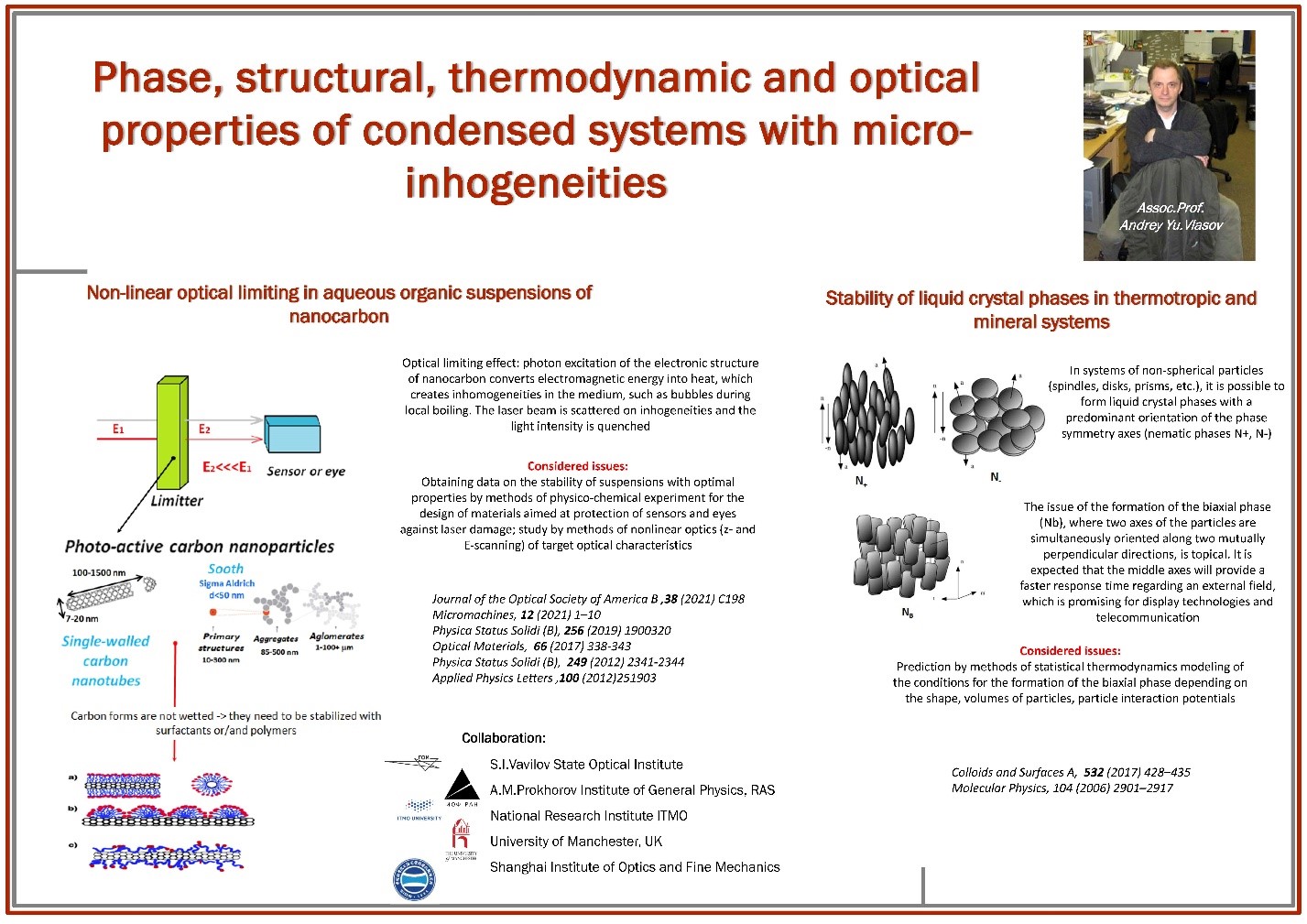
- Vlasov, A.Y., Venediktova, A.V., Ivanov, P.V., Nikolaeva, A.L., Anufrikov, Y.A. and Venediktov, V.Y. Aggregative Characteristics of Nanocarbon and of a Stabilizing Surfactant in the Aqueous-Polymer Matrix versus Optical Power Limiting Performance. Phys. Status Solidi B (2019) 256: 1900320. https://doi.org/10.1002/pssb.201900320
- Sofia Koroleva, Petr Korchak, Sabine Enders, Yuri Dobryakov, and Alexey Victorov. Solubility of n-Hexane and Setchenov’s Constants in Aqueous Solutions of KCl, NaCl, NaBr, and NaNO3. Journal of Chemical & Engineering Data (2019) 64, 10, 4314-4318 https://doi.org/10.1021/acs.jced.9b00384
- Anufrikov, Y. A., P. S. Kondrakhina, A. S. Koneva, and E. A. Safonova. Impact of Bioorganic Additives of Different Nature on Aggregation Behavior and on Cloud Point Temperatures of Nonionic Surfactants Tergitol NP-7 and Triton X-114 in Buffer Solutions. Colloid Journal (2019) 81 (6): 627-633 https://doi.org/10.1134/S1061933X19060024
- Ksenia A. Emelyanova, Alexey I. Victorov. Molecular thermodynamic modeling of a bilayer perforation in mixed catanionic surfactant systems. Phys. Chem. Chem. Phys. (2018), 20, 27924 - 27929 http://doi.org/10.1039/C8CP04593C
- Elena A. Belyaeva, Aleksandr A. Vanin, Alexey Victorov. Distribution of Zwitter-Ionic Tryptophan between the Micelles of 1-Dodecyl-3-Methyl Imidazolium and Aqueous Medium from Molecular Dynamic Simulation. Phys. Chem. Chem. Phys. (2018) 20, 23747-23753 https://doi.org/10.1039/C8CP02488J
- Yu. Gotlib, A. I. Victorov. Association equilibrium and gelation in solutions of cross-associating chains containing inactive fragments. Polymer Science, Series C (2018) Volume 60, Suppl. 1, 95–105 http://dx.doi.org/10.1134/S1811238218020091
- Alopina, E., Y. Dobryakov, E. Safonova, N. Smirnova, E. Kolobova, and L. Kartsova. Densities, Refractive Indices and Conductivities of Aqueous [Cnmim][Pro] Solutions (n = 4, 8, 12); Micellization and the Capillary Electrophoresis Data at 298.15 K. Colloids and Surfaces A: Physicochemical and Engineering Aspects (2018) 544: 137-143 https://doi.org/10.1016/j.colsurfa.2018.02.014
- Chernyshev, Y. S., Koneva, A. S. and Safonova E. A. NMR Self-Diffusion Study of Amino Acid Ionic Liquids Based on 1-Methyl-3-Octylimidazolium in Water. Applied Magnetic Resonance (2018) 49 (6): 607-618. https://doi.org/10.1007/s00723-018-1008-z
- Koneva, A. S., E. Ritter, Y. A. Anufrikov, A. A. Lezov, A. O. Klestova, N. A. Smirnova, E. A. Safonova, and I. Smirnova. Mixed Aqueous Solutions of Nonionic Surfactants Brij 35/Triton X-100: Micellar Properties, Solutes' Partitioning from Micellar Liquid Chromatography and Modelling with COSMOmic. Colloids and Surfaces A: Physicochemical and Engineering Aspects (2018) 538: 45-55 https://doi.org/10.1016/j.colsurfa.2017.10.044
- Kopanichuk, I.V., Vedenchuk, E.A., Koneva, A.S., Vanin, A.A. Structural properties of span 80/tween 80 reverse micelles by molecular dynamics simulations Journal of Physical Chemistry B (2018) 122(33), 8047–8055 https://doi.org/10.1021/acs.jpcb.8b03945
- Igor Gotlib, Alexey Victorov. Effects of the linear chain structure on the cross-association equilibrium between chainlike molecules in a good solvent. Fluid Phase Equil. (2017) V. 454. 116–121 https://doi.org/10.1016/j.fluid.2017.09.015
- Ksenia A. Emelyanova and Alexey I. Victorov. Driving Force for Spontaneous Perforation of Bilayers Formed by Ionic Amphiphiles in Aqueous Salt. Langmuir (2017) 33, N 46, 13438-13443 https://doi.org/10.1021/acs.langmuir.7b02885
- Ksenia Emelyanova, Igor Gotlib, Anna Shishkina, Michail Voznesenskiy, and Alexey Victorov Molecular Thermodynamic Modeling of Self-Assembly into Branches and Spatial Networks in Solution. Journal of Chemical & Engineering Data (2016), 61, (12), 4013−4022 https://doi.org/10.1021/acs.jced.6b00531
- Igor Yu. Gotlib, Ivan K. Malov, Alexey I. Victorov and Mikhail A. Voznesenskiy Association Equilibrium for Cross-Associating Chains in a Good Solvent: Crowding and other Non-ideality Effects, J Phys Chem B (2016) 120, 7234−7243 https://doi.org/10.1021/acs.jpcb.5b12530
- Ilia V. Kopanichuk, Alexandr A. Vanin, Igor Yu. Gotlib and Alexey I. Victorov Steric Asymmetry vs Charge Asymmetry in Dilute Solution Containing Large Weakly Charged Ions, Fluid Phase Equilibria (2016) 428, 203-211 http://doi.org/10.1016/j.fluid.2016.06.008
- Alopina, E. V., E. A. Safonova, I. B. Pukinsky, and N. A. Smirnova. Liquid-Liquid Equilibria in Aqueous Mixtures of Alkylmethylimidazolium Glutamate with Potassium Carbonate and some Physicochemical Properties of Aqueous [Cnmim][Glu] (n = 4, 6, 8) Solutions. Journal of Chemical and Engineering Data (2016) 61 (6): 2013-2019 https://doi.org/10.1021/acs.jced.5b00948
- Davletbaeva, P., M. Falkova, E. Safonova, L. Moskvin, and A. Bulatov. Flow Method Based on Cloud Point Extraction for Fluorometric Determination of Epinephrine in Human Urine. Analytica Chimica Acta (2016) 911: 69-74 https://doi.org/10.1016/j.aca.2015.12.045
- A I Victorov, M A Voznesenskiy, E A Safonova. Spatial networks in solutions of worm-like aggregates: universal behaviour and molecular portraits", RUSS CHEM REV (2015) 84 (7), 693–711 https://doi.org/10.1070/RCR4524
- A. Victorov. Molecular Thermodynamics of Soft Self-Assembling Structures for Engineering Applications. J Chem Technol Biotechnol. (2015) 90 (8), 1357-1363 https://doi.org/10.1002/jctb.4693
- S. V. Koroleva and A. I. Victorov. The strong specific effect of coions on micellar growth from molecular-thermodynamic theory. Phys.Chem.Chem.Phys. (2014) 16, 17422-17425 https://doi.org/10.1039/c4cp02178a
- Alexey I. Victorov. Modeling of Micelle-Solution Equilibria for Mixed Nonionic Micelles with Strong Specific Interactions in Coronae: Group-Contribution Approach. Journal of Chemical & Engineering Data (2014) 59, 2995−3002 https://doi.org/10.1021/je500103h
- Sofia V. Koroleva and Alexey Victorov. Modeling of the Effects of Ion Specificity on the Onset and Growth of Ionic Micelles in a Solution of Simple Salts. Langmuir (2014), 30, N 12, 3387−3396 https://doi.org/10.1021/la404845y
- Safonova, E. A., T. Mehling, S. Storm, E. Ritter, and I. V. Smirnova. Partitioning Equilibria in Multicomponent Surfactant Systems for Design of Surfactant-Based Extraction Processes. Chemical Engineering Research and Design (2014) 92 (12): 2840-2850 https://doi.org/10.1016/j.cherd.2014.04.005